Transient deletion of hypermutation preserves B cell affinity
by admin
Influence of an anti-DEC-OVA-induced G1-phase transition on the formation and size of human DNA helicase B cells
We compared the IgHV-7 to the IgHV-7 to see how affinity might impact formation and size. GC B cells that did not have or didn’t yet have one of the affinity-enhancers. Larger nodes were enriched among GC B cells carrying affinity-enhancing mutations (Fig. 2e). In conclusion, the experimental data are in keeping with the theoretical model suggesting that the GC reaction is optimized by mutation-free proliferative bursts of cells expressing high-affinity antibodies.
Intravital time-lapse imaging of actively cycling DZ B cells showed that, at 36 h after anti-DEC-OVA treatment, C/N ratios in DZ B cells remained steadily above the CDK2low G0 threshold (we were unable to track B cells for long enough to detect the increase in C/N ratios observed in vitro), whereas, in the absence of treatment, C/N ratios continued to decrease after mitosis, reaching the CDK2low threshold within 30 min of anaphase (Fig. 3l,m and Supplementary Videos 3 and 4). Thus, B cells actively undergoing inertial cycling in response to anti-DEC-OVA largely failed to enter a CDK2low G0-like state.
The G0 and G1 phases of the cell cycle have low activity of cyclin-dependency kinase 2 which is what promotes progression through G1 and S-phase initiation. To measure the phases in real life, we generated mice with a real-time reporter of CDK2. 3b,c). This reporter, based on previously published constructs43,44, consists of amino acids 994–1,087 of human DNA helicase B (DHB) fused N-terminally to the tdTomato red fluorescent protein and inserted into the ubiquitously expressed Rosa26 locus (Rosa26DHB-tdTomato; Fig. 3c). The nucleus is transferred to the cytoplasm after the CDK2phosphorylation. It is necessary to leave the nucleus when CDK2 activity is absent during G0, so that cells can progress through G1, S and G2 phases. 3b,d. The C/N ratio of DHB–tdTomato has been used to determine CDK2 activity, allowing for greater time resolution than can be achieved with conventional genetically-coded cells. The Histone 2B (H2B)-EGFP reporter47 was crossed to the B1-8hi B cell receptor and theTomato-td mice.
To experimentally test whether mutation rates are variable, and dependent on both affinity and division rates, we tracked GC B cell division in mice that express mCherry labelled Histone-2b (H2b-mCherry) under the control of a doxycycline (DOX)-sensitive promoter23,28. Lymphocytes from these mice (H2b-mCherry mice) constitutively express the mCherry indicator. Administration of DOX turns off the reporter gene and, upon dividing, cells dilute the indicator in proportion to the number of divisions made, whereas quiescent cells retain the indicator22,23,28 (Extended Data Fig. 1a–c.
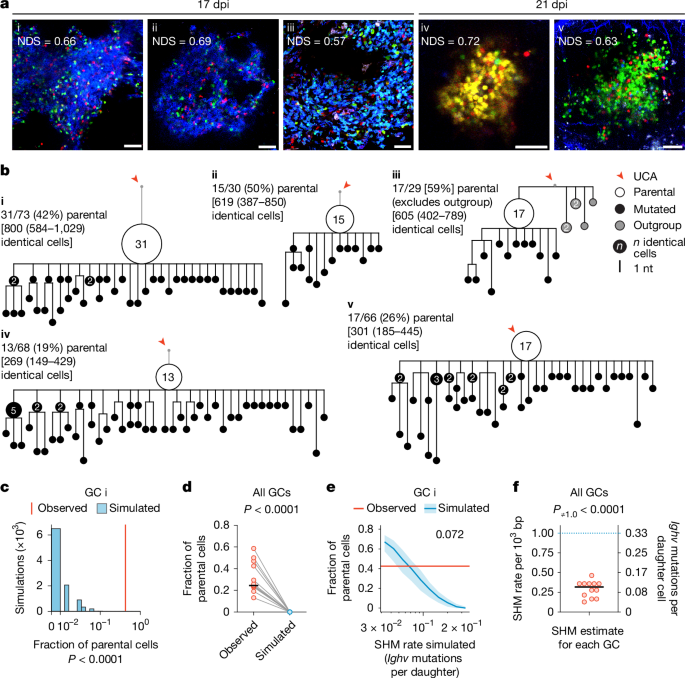
Our modelling predicts that a constant mutation rate of around 1 × 10−3 per base pair per cell division1, equivalent to approximately 50% chance of mutation per division pmut = 0.5, should produce branched trees containing limited small collections of identical sequences (nodes) of 15 or fewer cells (Fig. Black, 1c. Alternatively, the decreasing pmut model, with otherwise identical simulation parameters, predicted trees with nodes that extended up to 50 cells (Fig. 1c, red. Analysis of the experimental data revealed trees with nodes comprised of 2–15 members as well as trees with much larger nodes that contained 15–125 identical members (Fig. 2b and Extended Data Fig. 2f). The amount of nodes that could not be accounted for by the constant pmut model was more consistent with the decreasing pmut model.
Immune responses to simple haptens like NP might differ from more complex protein antigens. The analysis of GC B cells from H2b-mCherry mice that had undergone the vaccine was used to figure out the contribution of the clonal burst to immunization. After 14 days after vaccination and 36h after DOX exposure, GC B cells were barcoded according to RBD-binding andmCherry expression, allowing a pair of analysis of sequence identity, division status and RBD-binding as a surrogate for affinity. 3b). Genotype-collapsed phylogenetic trees were produced using paired IgH- and IgL-chain sequences from expanded clones (Fig. 3a,b and Extended Data Fig. 3c). The cells’ mCherry expression was annotated with the red and black outline of the RBD-binders. Similar to NP-OVA immunization, we observed large nodes containing the same sequence in which extensive Mutation-free clonal bursts occurred. When expanded clones were not considered, the fraction of all the nodes with more than 15 identical clones was less than half. When all cells were considered independently, there were an average of 25% of GC B cells derived from the same nodes with at least 15 identical sequence. 3d and Supplementary Table 5). The fraction of the nodes with more than 15 identical genes was always greater in the case of RBD+.
We were looking at how the constant pmut model could account for the large number of trees in the experimental data. To this end, we simulated the case with stochastic pmut (Extended Data Fig. 7a), and the case where the number of divisions corresponding to a constant level of T cell help was stochastic (Extended Data Fig. 7b). Both scenarios couldn’t account for the long-tail behavior of node sizes observed in experiments. Thus, both the theory and experimental data are consistent with the idea that the per division rate of SHM is regulated and decreases with increasing T cell help.
AID introduces C>U mutations at preferential nucleotide sequence hotspots (WRC, W = A/T, R = A/G, underline indicates residue targeted for mutation)37,38. To determine whether absence of SHM in expanded nodes was due to previous loss of these motifs, we profiled AID hotspots between cells contributing to nodes of sizes 1, 2–15 or more than 15. There were all three classes of cells that had hotspot motifs intact. 8d–f). Thus, target motif decay does not account for differences in SHM between nodes. The level of cell death among the mCherry compartments was found to be the same. 8g,h
Together these data are consistent with the idea that strong selection signals decrease the relative time DZ cells spend in G0/G1, thereby reducing their exposure to AID and lowering their per division mutation rates.
We showed that large clusters of MCherry-expressing Histone-2b (H2b-mCherry) cells are enriched in GC B cells carrying affinity-enhancing mutations. Here, we observed that DZ B cells remained above the CDK2Low G0 threshold at 36 h after anti-DEC-OVA treatment and C-N ratios in DZ B cells remained steadily above the CDK2low G0 threshold within 30 min of anaphase.